
One of the questions that came up when we were talking about dealing with the highly contaminated leachate that drains out of landfills, is what would happen to it if it was just put into a lake or the ocean. Would the liquid just mix into the water, or would it stay separate.
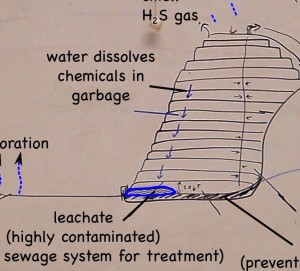
I’m afraid I did not go with an easy answer. It depends after all on two things: how different the density is of the leachate from seawater; and how turbulent is the water.
Turbulent water will make the leachate more likely to mix, while a greater density difference would cause them to “want” to remain separate. An extremely dense leachate might just settle to the bottom of a lake and stay there.
Small Islands
One example of two fluids that are in contact but stay separate is in the groundwater beneath small islands. Rain water falls on the island and seeps into the ground. It’s fresh, but the water in the surrounding ocean and the water that’s already underground are both salty. Salty water is more dense than the fresh so the freshwater will float on top of the salty water creating a thin lens.
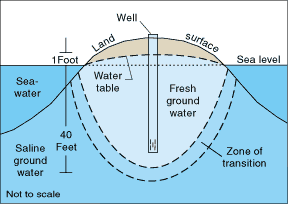
How thick is the lens? For every meter that the fresh groundwater is above sea level, there are 40 meters of fresh water below sea level (1:40). This is because saltwater has density of about 1.025 g/cm3, while freshwater has a density of about 1.000 g/cm3 (note that I use four significant figures in each of these values).
The freshwater lens can be a great source of drinking water on these isolated small islands, but like the islands themselves, they are threatened by rising sea levels due to global warming.