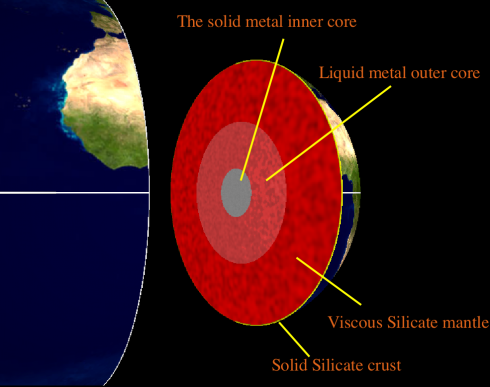
The inner core of the Earth is made of solid metal, mostly iron. The outer core is also made of metal, but it’s liquid. Since it formed from the solar nebula, our planet has been cooling down, and the outer core has been freezing onto the inner core. Somewhat counter-intuitively, the freezing process is a phase change that releases energy – after all, if you think about it, it takes energy to melt ice.
The energy released from the freezing core is transported upward through the Earth’s mantle by convection currents, much like the way water (or jam) circulates in a boiling pot. These circulating currents are powerful enough to move the tectonic plates that make up the crust of the earth, making them responsible for the shape and locations of the mountain ranges and ocean basins on the Earth’s surface, as well as the earthquakes and volcanics that occur at plate boundaries.

Eventually, the entire inside of the earth will solidify, the latent heat of fusion will stop being released, and tectonics at the surface will slow to a stop.
The topic came up when we were talking about the what heats the Earth. Although most of the energy at the surface comes from solar radiation, students often think first of the heat from volcanoes.
Note: An interesting study recently published showed that although the core outer core is mostly melting, in some places it’s freezing at the same time. Unsurprising given the convective circulation in the mantle.
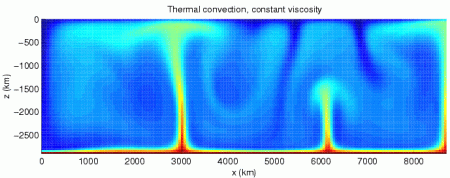
Note 2: Convection in the liquid outer core is what’s responsible for the Earth’s magnetic field, and explains why the magnetic polarity (north-south) switches occasionally. We’ll revisit this when we talk about electricity and dynamos.
