An interesting demonstration of how gold can be extracted from printed circuit boards (PCB’s) and RAM trimmings (which I did not know was a thing). The yield is low, but NileRed gives full detail, including the chemical equations, which is why I may show this to my chemistry class.
Tag: chemistry
Quick Chemical Formulas
While teaching chemistry this year, I’ve needed a quick way to write chemical formulas. The fastest thing to do is just write it out flat–sans sub and superscripts–like Na+, or CO2-. But that’s not pretty, and introduces another potential element that could confuse.
I found that you can use Lingojam’s sub and superscript generators to copy and paste the official UTF-8 characters for pretty much all the sub and superscripts I need for chemistry, but that takes a while to do.
So instead, I put together, just in time to write my finals, a little ChemEqn app that uses keyboard shortcuts to quickly substitute in the sub and superscripts as you type.
- [Ctrl][number]: give you the number subscripted:
- e.g. [Ctrl][2]: gives ₂
- [Ctrl][Shift][number]: gives superscripts:
- e.g. Ctrl][Shift][2]: gives ²
- [Ctrl][Shift][+]: gives a positive charge (⁺)
- [Ctrl][Shift][-]: gives a negative charge (⁻)
- [Ctrl][>]: gives a forward arrow (→)
- [Ctrl][<]: gives a backward arrow (←)
- [Ctrl][/]: gives the double arrow (⇌)
The method is not perfect, since you still have to decide which comes first when you have both a sub and superscript after an element (O₃²⁻ for example). Another issue is that when I do [Ctrl][Shift][+] and [Ctrl][Shift][-] it zooms into and out of the browser window since we’re using the default zoom shortcuts, but that’s, at least for me, a minor inconvenience.
I do like the app since it still makes for quite readable formulas that can be easily copied and pasted almost anywhere without messing up since it only uses UTF-8 characters that are pretty standard across the web (and most computer programs).
History of the Periodic Table
The first video is a quick overview of why Mendeleev’s table of elements was found to be so useful.
The second video goes into more depth about the periods and the groups.
Atom Board: Montessori Work

These atom boards worked very well for practicing how to interpret atomic symbols. The protons (blue) and electrons (red) are magnetic so they snap into place quite satisfyingly. Their poles are oriented so that the electrons will only attach properly to the slots in the electron shells and the protons only attach the right way up to the nucleus. The neutrons are wooden and non-magnetic.
Procedure for Building an Atom
Nucleus
Step 1: Number of protons (+ charge).
- The number of protons is given by the element name. Carbon will always have six protons, Hydrogen will have one proton. I have students memorize the first twenty elements in the correct order, so they can quickly determine the atomic (proton) number.
- 14C: Protons = 6+
Step 2: Number of neutrons.
- Neutrons = atomic mass – number of protons
- The atomic mass is given at the top left corner of the atomic symbol: 14 in the example above for 14C.
- 14C: Neutrons = 14 – 6 = 8
Electron Shells
Step 3: Number of electrons (- charge).
- Electrons = number of protons – charge
- The charge is given to the top right of the atomic symbol. In this case, there is no charge
- 14C: Electrons = 6 + 0 = 6
Step 4: Electron Shells
- Electrons go in shells around the nucleus.
- Start with the smallest shell, fill it, and then add the next shell until you’ve placed all of the electrons.
- The first shell can hold only 2 electrons, the second shell can hold 8, and the third 8. The electron configuration tells how many electrons are in each shell.
- 14C: Electron configuration: 2-4

They’ve also turned out to be useful when explaining ionic bonding. Since it’s easy to add or remove electron shells, you can clearly show how many electrons can be donated or received to figure out how many atoms are involved in the reactions.
Making 3D Periodic Tables

Ms. Fu’s chemistry class were given a project to make 3d periodic tables based on the properties of the elements. A few groups went with Makerspace options, using the new vinyl cutter and laser.


The part that took the longest was marking all the columns for cutting. A worthwhile assignment would be to write a program to automatically make the cut-marks in an svg file that can be etched with the laser.


Elements by Their Uses
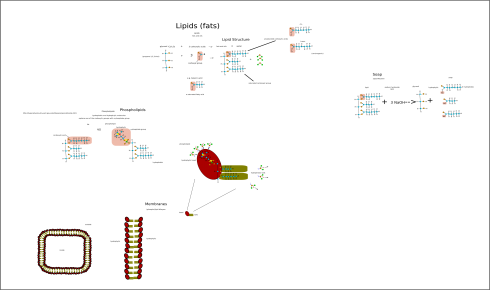
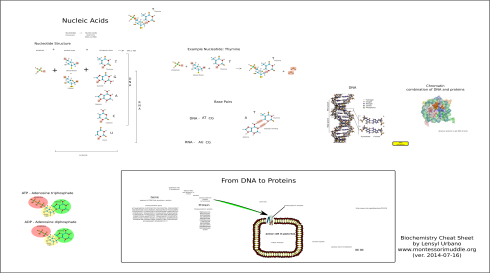
The Essential Biopolymers
Carbon in the Ground and Free Oxygen in the Air
A couple new article relevant to our study of Earth History.
Carbon
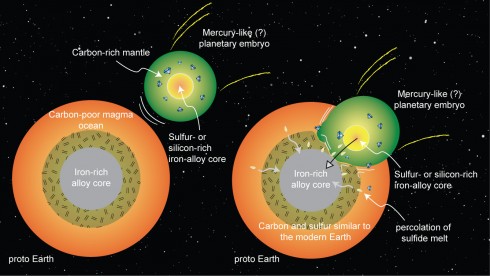
Research on the high pressure and temperature conditions at the Earth’s core suggest that most of the carbon in the early Earth should have either boiled off into space or been trapped by the iron in the core. So where did all the carbon necessary for life come from? They suggest from the collision of an embryonic planet (with lots of carbon in its upper layers) early in the formation of the solar system.
Free Oxygen
It took a few billion years from the evolution of the first photosynthetic cyanobacteria to the time when there was enough oxygen in the atmosphere to support animal life like us. Why did it take so long? NPR interviews scientists investigating purple microbial mats in Lake Huron.